Background: AZD0486 (formerly TNB-486) is a novel, IgG4 fully human CD19xCD3 bispecific TCE incorporating a unique low affinity anti-CD3 moiety designed to reduce cytokine release while retaining potent T-cell mediated cytotoxicity of malignant B cells. A silenced Fc prevents nonspecific binding, antibody-dependent cellular cytotoxicity, and confers a long half-life suitable for intermittent administration. Preliminary clinical activity in Diffuse Large B-cell Lymphoma (DLBCL) and Follicular Lymphoma (FL) was reported [ Hou et al, Blood 2022; Jacobs et al, HemaSphere 2023] following fixed and single step-up dosing (1SUD). We now present focused updated safety data following implementation of 2SUD in the ongoing FIH phase 1 study (NCT04594642).
Methods: Patients with R/R CD19+ B-NHL failing ≥2 prior lines of therapy, including prior CD19 CART and/or CD20 TCE, were enrolled. AZD0486 was administered in escalating doses in either no SUD (Day (D)1, D15 0.03-2.4mg), 1SUD (D1 0.27-1 mg, D15 2.4-10 mg), or 2SUD (D1 0.27 mg, D8 1 mg, D15 2.4-7.2 mg, FL/DLBCL only) schedules. AZD0486 was given IV every 2 weeks in 28D Cycles (C) up to 2 years. Monthly dosing was considered for patients in complete response (CR) at C6. Responses were assessed by RECIL 2017 by Central Imaging Review, AEs by CTCAE v5.0, and CRS/ICANS by 2019 ASTCT criteria.
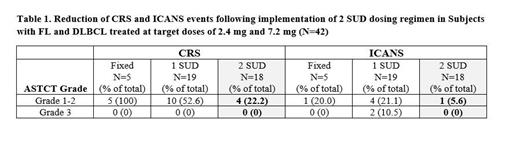
Results: As of 03 July 2023, 62 patients (27 DLBCL, 25 FL, 5 mantle cell lymphoma, 4 marginal zone lymphoma, 1 other) received AZD0486 and were evaluable for safety. Median age was 68 (range 22-86), with 58% male, 81% stage III/IV. Median prior lines of therapy was 3 (range 2-16) including 23 (37%) failing CART and 3 (4.8%) failing CD20 TCE. Fifteen (24%) were CD20 negative at study entry. The complete response (CR) rate was 91% (10/11) in patients with FL treated in 2.4 mg dose cohorts and for all FL efficacy evaluable subjects dosed at 2.4mg or above, CR was achieved in 14/17 (83%) with an overall response rate (ORR) of 88%. Median duration of response (DOR) has not beenreached. With the implementation of 2SUD regimen, no Grade 3+ CRS or ICANS events were observed (Table 1). Grade 1-2 CRS events decreased from 62.5% to 22.2% (fixed/1SUD, 15/24 and 2SUD, 4/18). Grade 1-2 ICANS events decreased from 20% (5/24) to 5.6% (1/18), presented mainly as confusion, and typically resolved in 1-2 days. The most common AEs (occurring in >20%) of any grade and regardless of drug causality were CRS (48%), anemia (34%), neutropenia (32%), lymphopenia (29%), ICANS (27%) and decreased WBC (20.9%). Treatment-related events of Grade ≥3 included lymphopenia (24%), neutropenia (15%), ICANS (9.7%) and anemia (3.2%). No treatment related deaths were reported (1 death due to COVID19, unrelated) and no treatment-related AEs lead to discontinuation (1 patient discontinued due to COVID-19, unrelated).
Preliminary analysis (TNF-a, IL-6, IL-8, IL-10) suggests a decrease in cytokine levels after target dose with 2 SUD compared to fixed dosing and 1SUD, regardless of target dose.
Conclusions: AZD0486 (formerly TNB-486) is an active treatment in patients with advanced R/R B-NHL and has a predictable safety profile characterized by mainly low-grade AEs and fully transient and reversible CRS/ICANS events. The 2 SUD schedule reduced the overall rate of low grade CRS and ICANS (Grade 1-2) and abrogated Grade 3 events further improving the risk/benefit profile of AZD0486. Dose escalation is ongoing to identify the RP2D.
Disclosures
Gaballa:TeneoBio: Research Funding; GiIead: Consultancy; BeiGene: Consultancy; AstraZeneca: Consultancy; ADC Therapeutics: Consultancy, Speakers Bureau; Ipsen: Consultancy, Research Funding; Genentech: Consultancy; Elli Lilly: Speakers Bureau. Jacobs:AbbVie: Consultancy, Research Funding, Speakers Bureau; Adaptive: Consultancy; Beigene: Consultancy, Speakers Bureau; Pharmacyclics: Consultancy, Research Funding, Speakers Bureau; Genentech: Consultancy; SecuraBio: Consultancy, Speakers Bureau; Teneobio: Research Funding; Janssen: Consultancy, Speakers Bureau; AstraZeneca: Research Funding, Speakers Bureau; LOXO Oncology: Research Funding. Devata:GSK: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months; Pfizer: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months; Eli Lilly: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months; Merck: Current equity holder in publicly-traded company; Johnson &Johnson: Current equity holder in publicly-traded company; AbbVie: Current equity holder in publicly-traded company; Bristol Myers Squibb: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months; Novo Nordisk: Current equity holder in publicly-traded company; Bayer: Current equity holder in publicly-traded company; AstraZeneca: Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Yoon:Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; Kirin Pharm: Honoraria, Speakers Bureau; Samyang: Research Funding; BMS: Honoraria, Speakers Bureau; Roche: Honoraria, Research Funding, Speakers Bureau; Boryung: Research Funding; Novartis: Consultancy, Honoraria, Speakers Bureau; Takeda: Honoraria, Speakers Bureau; Beigene: Consultancy; Pharos iBio: Consultancy; Abclon: Consultancy; GI cell: Consultancy; GC cell: Consultancy. Shah:BMS/Juno: Consultancy; Tundra Therapeutics: Current holder of stock options in a privately-held company; LOXO-Lilly: Consultancy, Other: Travel support; Epizyme: Consultancy; Janssen: Consultancy; Novartis: Consultancy; TG therapeutic: Consultancy; Umoja: Consultancy; Seattle Genetics: Consultancy; Gilead/Kite: Consultancy; Incyte: Consultancy; Abbvie: Consultancy; Lilly Oncology: Consultancy, Research Funding; Miltenyi Biotec: Consultancy, Other: Travel support, Research Funding. Brennan:AstraZeneca: Current Employment; Deciphera: Current equity holder in publicly-traded company, Ended employment in the past 24 months. Law:AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Chen:AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Sermer:AstraZeneca: Current Employment. Lesley:AstraZeneca: Current Employment, Current equity holder in publicly-traded company; Amgen: Current equity holder in publicly-traded company. Buelow:TBio, LLC: Current holder of stock options in a privately-held company; Ancora Biotech, Inc.: Current Employment. Forcina:AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Hou:AbbVie: Consultancy; AstraZeneca: Consultancy; Genentech: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal